(Portland, Ore.)—Niko Loening’s research is casting new light on proteins, one of the basic elements of all living species. Loening, assistant professor of chemistry, is an expert in protein-structure determination, and his work is raising questions about how proteins are formed and how proteins from one species may affect another.
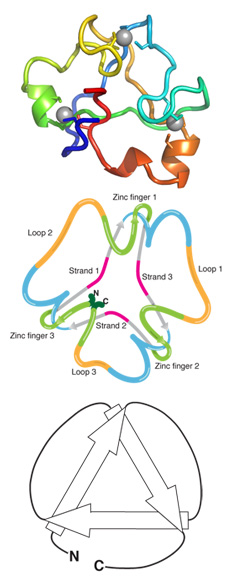
This summer, Loening published a study with colleagues in the UK, detailing a major discovery the group made: an unusual, knotted protein structure.
“Typically, the 3-D structure of proteins appears like a string folded in on itself,” he explained. “If you pull on both ends of most proteins, the protein strand unties like a shoelace. When you pull at both ends of the protein we discovered, instead of unraveling it tightens and forms a knot.”
As a protein’s structure determines its function, one major area of research over the past 50 years has been directed toward studying and predicting how strands of protein fold into 3-D structures. The discovery of this unique, knotted structure generates new questions about how proteins form.
“The structure that we determined in this project underscores how much we still have to learn about protein folding,” Loening said.
Beginning this fall, Loening will bring his expertise to bear on a different, consequential question, this time regarding proteins from brown recluse spiders. With the support of a grant from the National Institutes of Health, Loening will collaborate with Greta Binford, assistant professor of biology, to investigate the structure and function of proteins contained in spider venom toxins.
The $190,000 Academic Research Enhancement Award (AREA) Program Grant from the NIH will allow the team to engage in a three-year study of the peptides and proteins in brown recluse spider venom, which may have practical applications in the pharmaceutical industry.
“Brown recluse spider venoms contain hundreds of different toxins that target neurons and cause different reactions in victims—from injury to paralysis to death,” Loening said. “We want to study the specific effects of each of these toxins and we hope that, by determining their structures, we can gain some insight into how they operate. Since these toxins interfere with the nervous system, they can potentially be used in a controlled way to produce practical applications in medicine, such as a naturally derived, non-addictive pain reliever.”